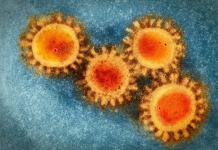
At the peak of the coronavirus, the FDA granted expedited use of different drugs and treatments in an effort to save lives. As more time has passed, they are looking at the data gathered over the last few months and are making some changes. Hydroxychloroquine, a drug made famous by President Trump’s endorsement of it during a speech, was thought to be an option for treatment. This week, however, the FDA has pulled approval for the drug to be used on COVID-19 patients. Per the data, there are too many risks and too few benefits, so it is no longer authorized for that use.
Read more on the story here
The revocation of emergency authorization also applies to another similar compound
The FDA is being accused of crushing small businesses
If you’re used to getting drug samples from your doctor, that may change
Malaria drugs are found not to be effective at treating the coronavirus